Lupin Pharmaceuticals Recalls 2 Lots of TydemyTM Due to OOS Results
31 July 2023 | Monday | News
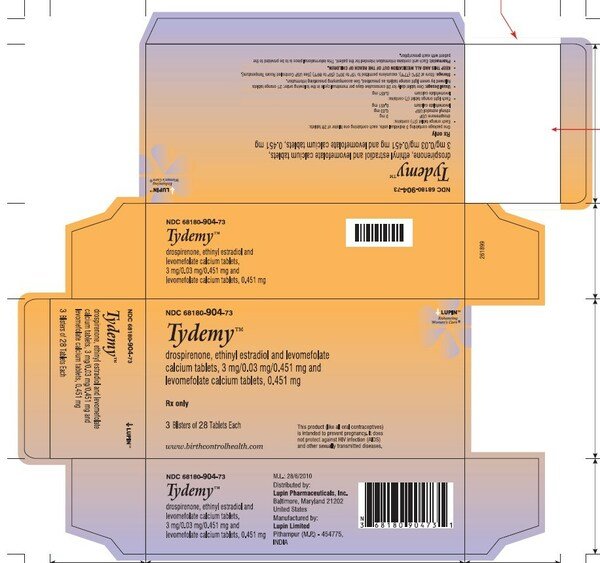
Lupin Pharmaceuticals Inc. (Lupin) is voluntarily recalling two (2) lots of Tydemy (Drospirenone, Ethinyl Estradiol and Levomefolate Calcium Tablets 3mg/0.03mg/0.451 mg and Levomefolate Calcium Tablets 0.451 mg) to the patient (consumer/user) level due to out of specification (OOS) test results at the 12-month stability time point. Specifically, one lot (L200183) tested low for ascorbic acid (an inactive ingredient) and high for a known impurity.
To date, Lupin has received no reports of adverse events related to either recalled lots. Regardless, Lupin is recalling two lots because if there were a significant reduction in the amount of inactive content (ascorbic acid), this could potentially impact the effectiveness of the product which could potentially result in unexpected pregnancy.
Tydemy is estrogen/progestin oral contraceptive (COC) indicated for use by women to prevent pregnancy and to raise folate levels in women who choose to use an oral contraceptive for contraception. Tydemy is packaged in 28's blister. One such blister was then packed in a pouch along with one printed sleeve, one pack insert (with day label) and one oxygen absorber (Stabilox) sachet. The three pouches were packed in one carton.
The lots were distributed nationwide in the US to wholesalers, drug chains, mail order pharmacies and supermarkets. The recalled lots are included in the table below:
|
Product |
Lot No |
Expiry |
NDC(s) |
UPC |
Distribution Dates |
|
TydemyTM |
L200183 |
Jan. 2024 |
68180-904-71 (1 Blister of 28 tablets each)
68180-904-73 (3 Blister of 28 tablets each) |
368180904731 |
June 2022 to May 2023 |
|
L201560 |
Sep. 2024 |
Lupin is notifying its wholesalers, distributors, drug chains, mail order pharmacies and supermarkets by phone and through recall notification and is arranging for the return of all the recalled product lots.
Patients taking, Tydemy are advised to continue taking their medication and immediately contact their pharmacist, physician, or medical provider for advice regarding an alternative treatment.
Wholesalers, distributors and retailers that have Tydemy that is being recalled should discontinue distribution of the recalled product lots immediately.
Consumers, wholesalers, distributors, and retailers with questions regarding this recall should contact Inmar Rx Solutions, Inc. at (866) 480-8206 Monday – Friday 09:00 am to 05:00 pm EST. For reimbursement, please have the recalled lots returned to Inmar Rx Solutions, Inc.; the lot number can be found on the side of the carton. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178.
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks




