Samsung Biologics continues to leverage remote audits and regulatory inspections
24 June 2021 | Thursday | News
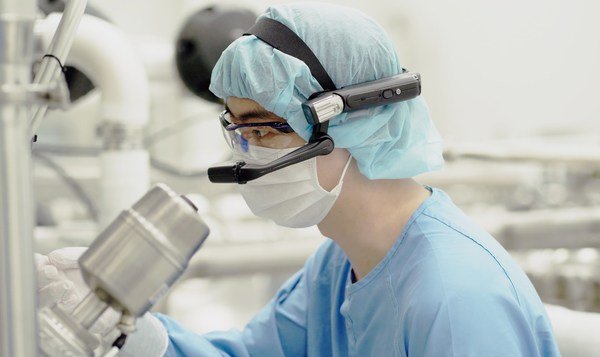
Samsung Biologics Live Virtual Tour
- Samsung Biologics' Live Virtual Tour (LVT) used in one of the earliest FDA full virtual assessments
- Recently hosted EMA for 6 PAIs simultaneously using LVT
Samsung Biologics, a global contract development and manufacturing organization (CMDO), celebrated the one-year mark of Live Virtual Tour (LVT), a real-time interactive platform to provide remote site inspections for both clients and regulatory agencies. The innovative digital solution was developed and implemented at Samsung Biologics a year ago in the wake of the pandemic and has since been successfully utilized in over 50 virtual regulatory and client audits and inspections and counting.
When many regulatory agencies and clients had postponed or completely discontinued GMP site inspections due to COVID-19 travel restrictions, Samsung Biologics proactively leveraged its process innovation to custom tailor a robust solution specifically to address the rigorous demands of a biopharmaceutical onsite inspection. Live Virtual Tour is an omni-device platform which allows third parties, such as the FDA and clients, to remotely observe and assess Samsung Biologics' facilities, communicate in real-time, and review GMP documentation, all in high definition using a flexible and secure global cloud infrastructure.
Since the launch of LVT, the company has utilized the platform for many aspects of its business, including technical due diligence and regulatory inspections including one of FDA's earliest virtual assessment for Emergency Use of Authorization (EUA) of COVID-19 treatments, and more recently, EMA's virtual inspection ("EMA distant assessment") for six Pre-Approval Inspections (PAI) over the course of seven days, receiving no critical observations.
John Rim, CEO of Samsung Biologics, said of the inspections, "While we have had many successful regulatory inspections in the past, these recent approvals, are meaningful milestones for Samsung Biologics. We will continue to adopt new technologies and stay abreast of what regulatory authorities require to help our clients secure robust and safe delivery of treatments to all patients around the globe."
The FDA issued a new recommendation in April providing guidance on remote inspections ("Remote Interactive Evaluations") in light of COVID-19, suggesting that the FDA may continue to use remote inspections to supplement on-site audits post-pandemic. The European Federation of Pharmaceutical Industries and Associations (EFPIA) also predicts added value in retaining a virtual inspection system beyond the pandemic.
By using all available approaches to ensure drug products are safe, effective, and of high quality, regulatory agencies agree that this form of evaluation is an effective and adaptive strategy for the duration of the COVID-19 public health emergency. James Choi, Samsung Biologics Senior VP and Chief Information and Marketing Officer said, "Our Live Virtual Tour platform not only fully complies with the regulatory agencies' guidance on remote interactive evaluations but is also aimed at promoting greater client satisfaction through digital and virtual access to our site and quality documents."
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks




