DeepQure Initiates Early Feasibility Study for Groundbreaking HyperQure™ System Following FDA Approval
11 June 2024 | Tuesday | News
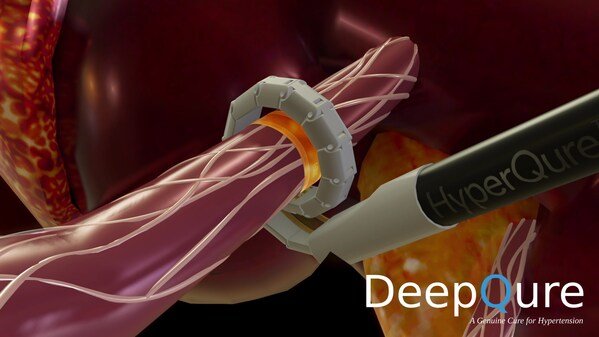
Image Source : Public Domain
DeepQure, a Seoul based medical device company with a novel, extravascular (laparoscopic approach) solution for renal denervation (RDN), announced initiation of its Early Feasibility Study (EFS) for the HyperQure™ system following FDA IDE approval. HyperQure™ is the world's first extravascular RDN medical device for the treatment of resistant hypertension.
With this approval, the company will commence the Early Feasibility Study to prove the safety and efficacy of HyperQure™ in 15 patients with resistant hypertension. The clinical trial will be conducted in a prospective, multicenter, single-arm, open-label design at major U.S. university hospitals, including Stanford University, Mayo Clinic, Emory University, University of Arizona and the University of California, Irvine.
"We are thrilled that the FDA has approved our IDE study plan. This is a significant US regulatory milestone for DeepQure Inc., starting the feasibility study using the extravascular ablation platform in the US for the Renal Denervation indication. We will accelerate our global clinical trials with this IDE approval," says Chang Wook Jeong, co-founder and CMO of DeepQure Inc.
HyperQure™ system consists of an energy generator and a laparoscopic instrument that delivers RF energy for ablation directly to the sympathetic nerves around the renal artery by wrapping the renal artery 360 degrees from outside of the vessel. This mechanism allows full denervation of renal sympathetic nerves without damaging the vascular endothelium which has proven to be a challenge for intravascular (catheter) systems. The HyperQure system is a simple, straightforward device that is easy to learn and aims to deliver uniform denervation results regardless of the surgeon's skill sets.
DeepQure Initiates Early Feasibility Study for Groundbreaking HyperQure™ System Following FDA Approval
Most Read
- How Does GLP-1 Work?
- Innovations In Magnetic Resonance Imaging Introduced By United Imaging
- Management of Relapsed/Refractory Multiple Myeloma
- 2025 Drug Approvals, Decoded: What Every Biopharma Leader Needs to Know
- BioPharma Manufacturing Resilience: Lessons From Capacity Expansion and Supply Chain Resets from 2025
- APAC Biopharma Review 2025: Innovation, Investment, and Influence on the Global Stage
- Top 25 Biotech Innovations Redefining Health And Planet In 2025
- The New AI Gold Rush: Western Pharma’s Billion-Dollar Bet on Chinese Biotech
- Single-Use Systems Are Rewiring Biopharma Manufacturing
- The State of Biotech and Life Science Jobs in Asia Pacific – 2025
- Asia-Pacific Leads the Charge: Latest Global BioSupplier Technologies of 2025
- Invisible Threats, Visible Risks: How the Nitrosamine Crisis Reshaped Asia’s Pharmaceutical Quality Landscape
Bio Jobs
- Sanofi Turns The Page As Belén Garijo Steps In And Paul Hudson Steps Out
- Global Survey Reveals Nearly 40% of Employees Facing Fertility Challenges Consider Leaving Their Jobs
- BioMed X and AbbVie Begin Global Search for Bold Neuroscience Talent To Decode the Biology of Anhedonia
- Thermo Fisher Expands Bengaluru R&D Centre to Advance Antibody Innovation and Strengthen India’s Life Sciences Ecosystem
- Accord Plasma (Intas Group) Acquires Prothya Biosolutions to Expand Global Plasma Capabilities
- ACG Announces $200 Million Investment to Establish First U.S. Capsule Manufacturing Facility in Atlanta
- AstraZeneca Invests $4.5 Billion to Build Advanced Manufacturing Facility in Virginia, Expanding U.S. Medicine Production
News
Editor Picks




